The Institute for Translational Sciences (ITS) Informatics Team now providing an updated Research Data Request Service. The service is available to investigators for pre and post award activity including pilot projects supporting future
grant applications. The request process provides for an Investigator to request free access to deidentified data or a fee based service for investigators requiring identifiable data. Scroll down to view workflow.
De-identified patient data is
available through ACT/SHRINE or TriNetX applications!
All UTMB users can request access to TriNetX or the ACT/SHRINE application! The de-identified EMR dataset makes it permissible for any UTMB user to easily obtain access and perform
searches.
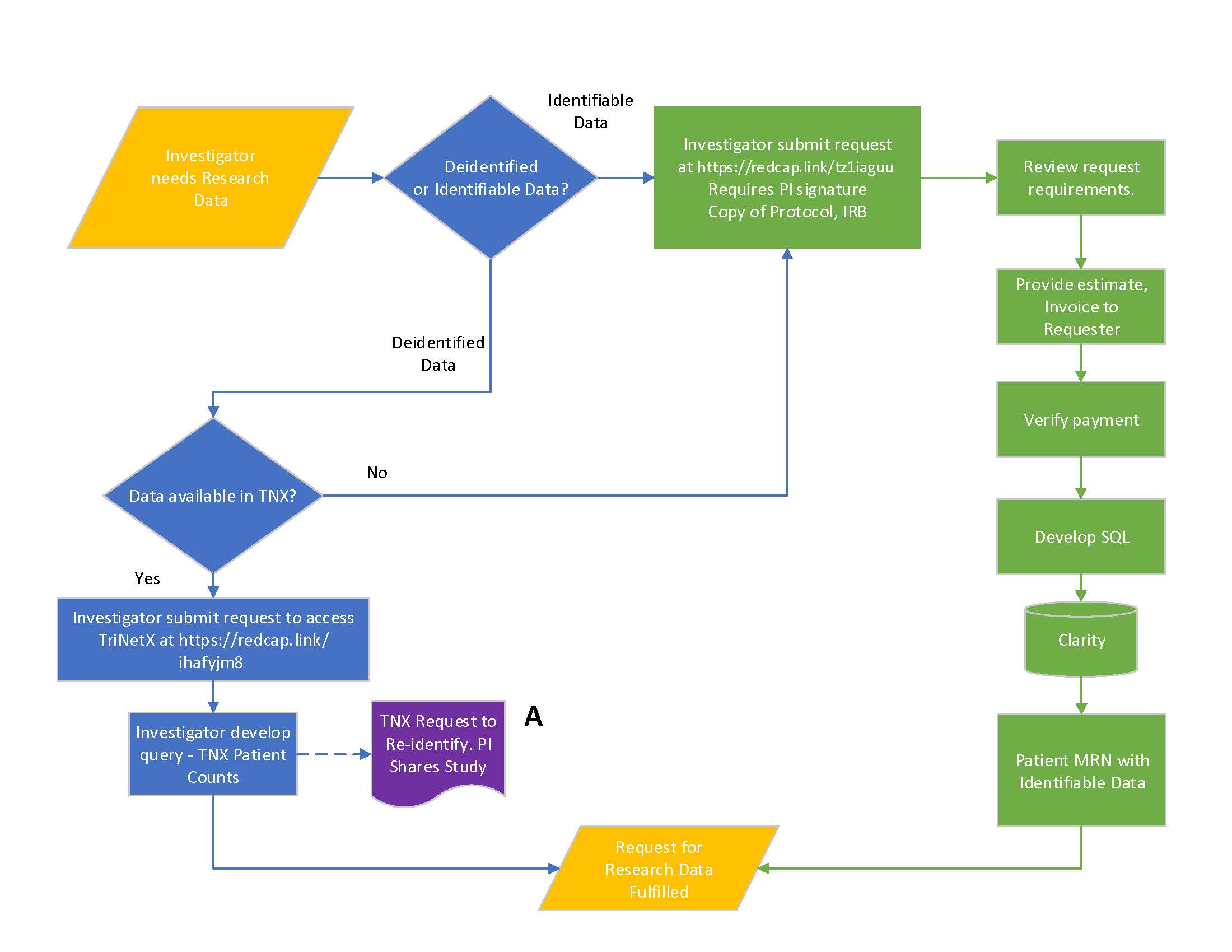
Research Data Request Procedure:
- Investigators are responsible for including sufficient details in a study protocol that describes the type of data needed to perform their research, whether it will be identified or deidentified, and the data analysis plan. The updated request procedure
divides requests into two specific workflows depending on type of data requested.
- For deidentified data, investigators can go to https://redcap.link/ihafyjm8 and submit a request to
access i2b2, ACT/SHRINE, or TriNetX. Investigators will have the capability to create and run queries against deidentified data using either application. Training is available.
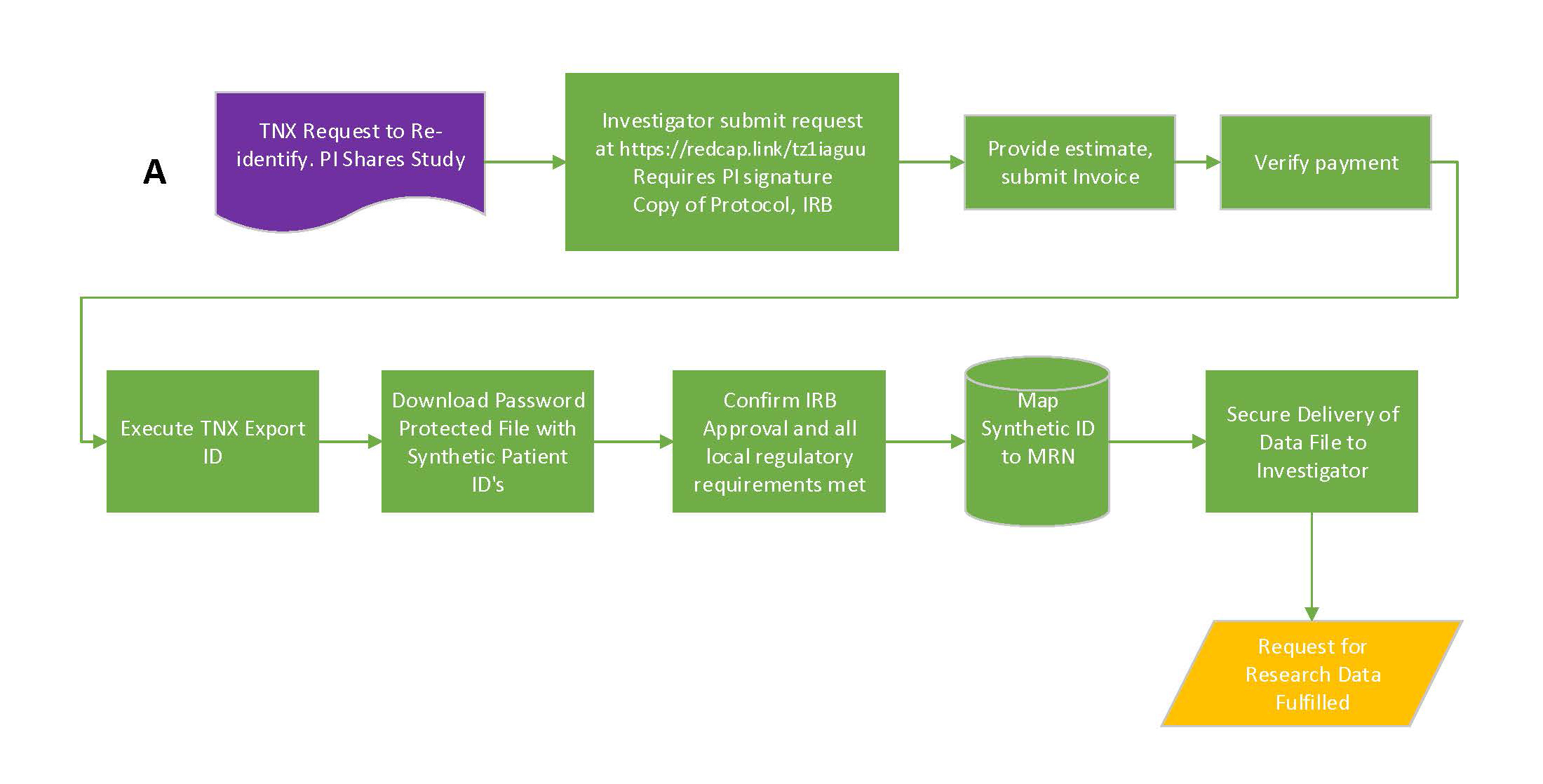
Investigators using TriNetX, with IRB approval, can submit a request to re-identify patient IDs found in TriNetX to UTMB MRN’s. This type of request is supported as a request for identifiable data and governed by the process below.
- For identifiable data, investigators can submit a research data request at https://redcap.link/tz1iaguu.
Investigators are responsible for submitting one of the following when requesting identifiable data:
o IRB approval for the study protocol
o IRB exemption for the study protocol
o IRB confirmation that the research does not constitute Human Subject Research
- ITS will review the research data request, protocol, and IRB memo to ensure that the data request is consistent with the protocol and IRB memo.
- ITS will provide a complimentary one hour consult to review the data request with the Investigator. Additional information may be requested if the request is unclear or inconsistent with the study protocol and/or IRB memo. The fee for Research Data
Requests are $100.00 per hour following the complimentary consult.
- Investigators will provide clarification and, if needed, additional documentation to support the IRB approval for the requested data.
- Failure by the Investigator to provide sufficient clarification or documentation will result in the data request being declined or withdrawn by ITS.
- ITS will provide an invoice to the data requester’s department administrator for the actual data request support hours required to provide the requested data following the initial consult. ITS will track the number of actual hours o the invoice
with the requester before delivering the requested data.
- Upon receiving payment, the data will be provided to the requester in accordance with the request, protocol, and IRB memo. Under no circumstances will additional data be provided outside the parameters of the request, protocol, and IRB memo.
- If the Investigator needs additional data or data that falls outside the scope of the protocol and IRB memo, the Investigator is responsible for amending the protocol and obtaining additional IRB approvals. The Investigator will provide these documents
with a new data request submitted to ITS.
- UTMB data provided to an Investigator may not be shared with non-UTMB personnel or non-UTMB entities unless a fully executed agreement t is in place between UTMB and the non-UTMB personnel or entities. NOTE that Investigators do NOT have signatory
authority to execute such an agreement and only designated UTMB signatories may sign an agreement.
Contact Gerald Brown at ge3brow@utmb.edu if you need more information.
Who can use it? The service is available for UTMB Investigators supported by or leading to external funding.